Why we need plutonium power for space missions

Written by
Casey Dreier
Chief of Space Policy, The Planetary Society
August 17, 2021
All spacecraft need electrical power to function. Most use solar panels that harvest energy from the Sun, but this solution has its limitations. Missions exploring the distant reaches of the solar system cannot generate enough energy from the distant, dim Sun. Shadowed craters, two-week-long lunar nights, and the dusty plains of Mars also prevent dependence on solar energy for long-lived missions. We need another source of power to explore these extreme cosmic locales.
That solution is Plutonium-238 (Pu-238), a non-weapons-grade radioactive material that generates large amounts of heat. This heat is used to generate electricity for spacecraft day or night, dusty or clear, distant or not. It powers the Voyager spacecraft beyond the solar system, Perseverance on Mars, and Cassini during its decade-long exploration of Saturn.

Pu-238 does not occur naturally in useful amounts. It must be created via an advanced and specialized industrial system — a mainstay of modern space infrastructure that must be constantly maintained and supported in order to provide access for our most advanced spacecraft.
The United States stopped making Plutonium-238 in 1988, when changes in nuclear weapons policy closed down the single production facility in the country. It wasn’t until 2014 that the Department of Energy, working on behalf of NASA, created the first new grams of this critical material. Current plans are to increase the scope of production to 1.5 kilograms (3.3 pounds) per year by the mid-2020s — enough to meet NASA’s current scientific needs.
The Planetary Society strongly supports the safe production and use of Plutonium-238 for scientific and exploration purposes. The U.S. stockpile must be constantly replenished in order to maintain useful amounts for future space missions, and The Planetary Society considers the ability to produce Pu-238 as enabling infrastructure for planetary exploration.
How Plutonium-238 Creates Energy
Heat can be converted into electricity using a device known as a thermocouple. Anything that gives off excess heat can fuel a thermocouple, to varying degrees. Plutonium-238 is extremely hot and dense with energy — enough to power spacecraft for decades at a time. No other material is comparable.
The heat from Plutonium-238 arises from its natural radioactive decay into Uranium-234. As Pu-238 decays, it emits small particles composed of two protons and two neutrons. These are relatively hefty particles that careen and crash into other nearby atoms in the Pu-238 fuel, creating movement and thermal energy. This results in temperatures of roughly 1,350 degrees Celsius (2,462 degrees Fahrenheit).
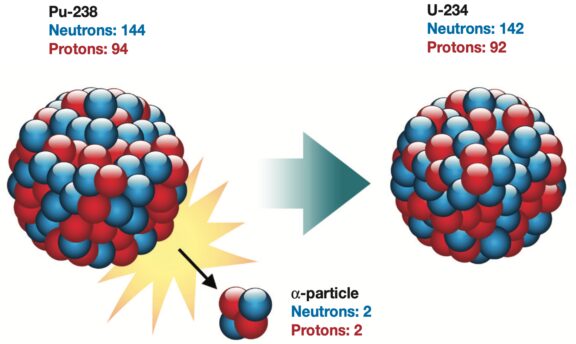
To convert the heat from Pu-238 decay into energy, spacecraft use a type of thermocouple called a radioisotope thermoelectric generator. RTGs are cylindrical devices with fins that can be seen sticking off the side of spacecraft. They have no moving parts and are extremely rugged and reliable, working for decades at a time.

Spacecraft can also use the excess heat created by Plutonium-238 decay to warm sensitive components, reducing the need for power-hungry electrical heaters. Even missions that primarily depended on solar panels for energy, such as the twin Mars Exploration Rovers, sometimes include small pellets of Pu-238 to provide passive heating that helps extend the life of the spacecraft.
Making Plutonium-238
Plutonium-238 poses a problem: it is constantly disappearing. Unlike fission nuclear power, where energy production can be regulated, Pu-238 decays at a constant rate. This rate is known as an element’s half-life, the amount of time it takes for half of any given radioactive substance to change to its stable atomic state. For Plutonium-238, it’s 88.7 years. This is long enough to be useful for space exploration missions, but short compared to geologic timescales. Any naturally occurring Pu-238 decayed eons ago, leaving only trace amounts to be found on Earth. Therefore, Plutonium-238 must be created. And not just once, but constantly, in order to replace the constant amounts lost to its unceasing decay.
Creating Plutonium-238 is not easy. It requires nuclear reactors, an advanced industrial base, and skilled engineers and technicians. It is also radioactive, and therefore subject to intense health, safety, and security protocols.
During the Cold War of the mid-20th century, large amounts of Plutonium-238 were created as a byproduct of nuclear weapons production. But as the nuclear arms race wound down in the late 1980s, the prime source of production, the Savannah River Site in South Carolina, was shut down. This left the United States with an existing but gradually depleting stockpile of useful Pu-238 for space exploration.
The Department of Energy produces all radioactive materials for the United States, for medical needs to nuclear bombs. NASA cannot make its own plutonium; it relies on the DOE.

In the early 2000s, after decades of decay, the national stockpile of useful Pu-238 was dwindling to crisis levels. Individuals at NASA and organizations such as The Planetary Society worked to secure funding to restore production capability of Plutonium-238 for space missions.
This was not merely a switch that could be turned on. After the closing of Cold War-era facilities in the 1980s, new methods and new production techniques had to be developed and validated at existing DOE laboratories throughout the country.
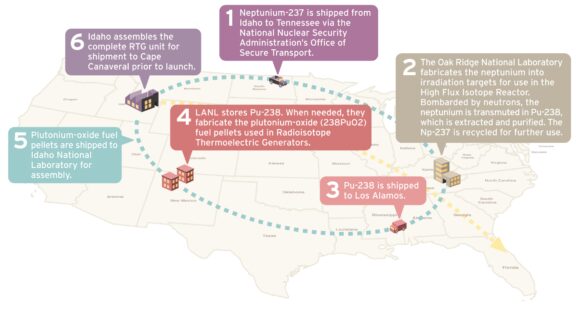
To make Plutonium-238, technicians start with Neptunium-237, an abundant byproduct of nuclear reactors. Neptunium-237 is irradiated in one of two research reactors maintained by the Department of Energy, and then purified and mixed with oxygen to create a rigid ceramic form of Plutonium-238 fuel used inside of RTGs. This process utilizes three different DOE facilities scattered throughout the country and takes years to complete.
Starting in 2014, NASA began reimbursing the Department of Energy for its overhead costs relating to Pu-238 generation, storage, and RTG production. The agency’s Planetary Science Division now pays approximately $150 million per year for this service.
By 2024, the DOE plans to produce 1.5 kilograms (3.3 pounds) of Plutonium-238 dioxide per year, enough to fuel a full RTG every 3-4 years. While it sounds small, this is enough to meet NASA’s current needs for space missions through the 2020s.
Safety
Though radioactive, Plutonium-238 is not as dangerous as some types of radioactive isotopes. The comparatively beefy alpha particles it emits are easily blocked by materials as thin as paper, though the element itself is shielded by far studier stuff when placed inside an RTG. The danger comes from inhalation rather than surface exposure.

To mitigate this, Plutonium-238 fuel is produced in a study ceramic form which resists shattering and is insoluble in water. This fuel is then encased in multiple layers of graphite and other study materials, designed to withstand the forces associated with a rocket explosion. The resulting system is so resilient, that after an aborted launch of a plutonium-powered weather satellite in 1968, the RTG unit was recovered on the ocean floor and re-used on a subsequent mission.
Nevertheless, launches that include Pu-238 are subject to extensive environmental and security reviews, which add tens of millions of dollars to the cost of the mission. Ultimately, the president of the United States approves most launches with radioactive material.
An Ongoing Concern
The Planetary Society advocated for years to restore Plutonium-238 production in the United States. It is a great success story for space advocates, scientists, and exploration that new Pu-238 is actively being produced and readied for use in new missions. As of today, its availability is not a major concern.
But because Pu-238 never stops decaying, we must never stop producing it to maintain our supply. It is an ongoing and permanent necessity of space infrastructure. Plutonium power enables the missions to dramatic locales: deep space, lands of shadow and dust such as Martian caves, ice-rich lunar craters, the hazy skies of Titan, and more.


 Explore Worlds
Explore Worlds Find Life
Find Life Defend Earth
Defend Earth

