Capturing the Rhythm of Space: Insights from 47th DPS Meeting

Written by
Deepak Dhingra
Postdoctoral Fellow, Department of Physics, University of Idaho
January 7, 2016
Space represents a domain of infinite possibilities ranging from subtle processes to the most dramatic events—each of them playing their role in composing the symphony that we all are trying to understand, appreciate and relish! The Division of Planetary Science (DPS) Meeting held in November saw many exciting scientific discussions spanning the range of processes on different planetary bodies, as well as their replication in the laboratory and in models. I was particularly intrigued by the rich diversity in the studies of planetary ices and share the details of some of them here.
Josh Colwell from University of Central Florida discussed results from what he refers to as the NanoRocks experiment. It aims to help further our understanding of the collisional evolution of particles in the planetary rings. He and his team are conducting experiments in microgravity aboard the International Space Station (ISS) at very low velocities (~ 1 mm/second) between a variety of grains to simulate collisions between the particles in planetary rings. The innovative experiment is comprised of grains of different sizes (0.1–2 mm) and composition (plastic beads, copper, glass and lunar soil simulant) housed in 8 compartments. The entire sample container is gently shaken at 1 minute intervals to achieve velocities of few centimeters per second. The collisions between the particles in all the compartments are monitored using a video camera in order to record how their velocities and trajectories change over time.

These experiments are helping Josh and his team to estimate a parameter called the "mean coefficient of restitution." This is essentially a measure of kinetic energy available for particle rebound after a collision and the amount of energy lost in the collisions. The team uses an image processing based method to derive this parameter. The method has been validated by an independent (and laborious) methodology of tracking individual particle trajectories. In the case of the image processing method, several frames obtained over a short time period are averaged and subtracted from another set of averaged frames taken later in time to estimate the velocity dispersion or the spread of velocities. Over time, the collisions are expected to dampen particle velocities.


Josh explained that the evolution in the velocity distribution is independent of particle collision velocities. This is in agreement with experiments carried out with larger ice particles by other researchers. The experiment is also relevant in another much broader context— namely, how small particles stick together to form bigger particles, giving rise to planetesimals that eventually form planetary bodies. It has been a challenge so far to understand this phenomenon. The NanoRocks experiment found the onset of cluster formation between particles at velocities of a few mm per second. Josh mentioned that work remains to be done on several image frames that have been obtained so far, and it will be a while before they are able to fully characterize this rhythm of collisional interaction between small particles.
Shifting gears, let's discuss the character of water ice—something that I have also started working on recently. Water ice can take on either an "amorphous" (i.e., lacking a crystal structure) or "crystalline" character during its formation. There are many flavors of ices, as shown in the image below. Some of these are better-known than others. One area of research in this field is the attempt to discretely define the boundaries between the various types of ices. Emma Mitchell from University of Virginia, Charlottesville touched upon one such mystery: the occurrence of crystalline ice at temperatures where we'd typically expect to see amorphous ice.
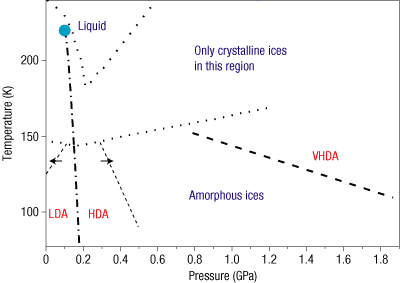
Emma presented experimental evidence for the role of microporosity—empty space between adjacent grains—in the formation of crystalline vs. amorphous water ice. Using a sophisticated set of experiments, she deposited thin films of amorphous water ice from vapor at 10 K. The angle with respect to the surface on which the thin film was deposited varied in experiments from 0–70 degrees. This angle is related to the amount of porosity attained by the water ice samples. The samples were then heated to 130–140 K to observe the transition of amorphous ice to crystalline ice, and to compare the trends between different flavors of amorphous ices. Emma showed that amorphous ice formed at deposition angle of 70 degrees transitioned to crystalline ice at a rate that was ~14 times faster than in the case of amorphous ice deposited at 0 degrees. In terms of differences in the porosity of these two types of amorphous ice, preliminary estimates indicate ~2.5x higher porosity for ice deposited at 70 degrees angles. Thus, it seems that higher porosity amorphous ice could rapidly transition to crystalline ice, making it more common at lower temperatures than is generally expected. Emma continues to refine her estimates. I eagerly await her publication. :)
In case you're wondering about the possible scientific implications for finding crystalline vs. amorphous ice, I could chip in with my two cents (a little bit of an advertisement!). At the conference, we presented our work focusing on the remote-sensing-based analysis of water-ice particles in Enceladus’ plume. We are consistently finding that these water ice particles are crystalline in nature, which implies that they formed at warmer temperatures (likely above 130 K). Since the water-ice particles are samples from the interior of Enceladus and are being observed almost instantaneously, the predicted warmer temperatures are representative of the subsurface temperatures where the water-ice particles are being formed. That’s an important insight. In addition, the higher temperature prediction also has a bearing on the likely formation process for these water-ice particles.
Continuing our dialog of the planetary ices, let's now jump to spacecraft observations of icy moons. However, the focus here is on components that are non-icy (i.e., rock). It has long been speculated that several of the icy moons—including Europa, Enceladus and Iapetus—also have non-icy components on their surfaces. However, the precise detection of these components has proven to be a hard task due to several complexities, including reproducing the environments that likely exist at these moons. From an observational standpoint, the instrument capabilities are, at times, not well-suited to identify the spectral signatures of predicted components. And sometimes there are just too many possibilities that could explain the observed signatures.
Cindy Young from Georgia Tech and her colleagues tried approaching this problem by looking at the icy bodies at a different wavelength range than is conventionally used. She utilized data from Composite Infrared Spectrometer (CIRS) instrument onboard Cassini to probe the signatures of icy moons in the mid-infrared range (600–1400 cm-1 in the context of CIRS measurements). Her work has led to the identification of an emission spectral feature at 855 cm-1 along with a possible doublet feature around 660 and 690 cm-1. These features originate from the enigmatic dark region of Iapetus, one of Saturn’s icy moons. Cindy highlighted the fact that due to insufficient measurements of silicates under conditions prevalent on the icy moons, unique identification of a spectral signature is a challenge. This is because the spectral signature of materials can vary depending on the ambient conditions. However, based on their laboratory measurements under vacuum and at 125 K, muscovite (a clay mineral) is the closest match to the observed spectral signatures on Iapetus, making it a strong candidate. However, more work is required to tie this detection uniquely to muscovite. This is however the first time that spectral features have been isolated based on CIRS observations of Saturn's icy moons. In addition, a strong case has been made for the detection of silicates on icy bodies, which has been speculated for a long time. It’s an important finding since the composition of the dark material would provide further clues to its origin and possibly the surface evolution of icy bodies over time.
The astounding diversity of planetary processes across the solar system collectively defines the rhythm of space. Which component of this rhythm resonates with you?


 Explore Worlds
Explore Worlds Find Life
Find Life Defend Earth
Defend Earth


