Paul Hayne • Jan 16, 2014
Dry Ice Snowfall at the Poles of Mars
Our exploration of the Solar System is often intertwined with the search for life, which in turn is a search for conditions – past or present – similar to those of our home world. With its abundant river channels, hydrated minerals, and ancient lakebeds, Mars strikes many as the most Earthlike planet in the Solar System (after our own, of course). Even its glistening white polar ice caps are reminiscent of Earth. Yet as the Red Planet continues to surprise us with the familiar, it is also host to some of the most bizarre and fascinating phenomena in the planetary neighborhood.
Let's take a closer look at those polar caps. With a large enough telescope, you can observe their seasonal growth and retreat, just as scientists did in the late 19th century. By analogy with Earth, they were first presumed to be composed of water ice, until a pair of researchers at Caltech in the 1960s showed that winter temperatures should drop to a bitter –125°C. At that point, the Martian atmosphere itself (composed primarily of carbon dioxide, CO2) should begin to freeze out. Thus, the seasonal ice caps of Mars ought to be composed primarily of CO2, rather than water ice. Given that no spacecraft had ventured beyond Earth orbit at the time, Robert Leighton and Bruce Murray's model was surprisingly accurate, and many of their predictions were borne out by planetary spacecraft observations in the following decade.
One of the most striking predictions of the Leighton and Murray (LM) model was that over the Martian seasons the mass of the atmosphere should vary by ~30% as CO2 is exchanged with the polar caps. LM therefore showed that this atmospheric freeze-out should be expressed in pressure readings taken anywhere on the planet. When the Viking landers touched down in 1976, it had been ten years since LM’s publication. As the pressure data came in, the telltale signature of the Martian global CO2 cycle showed up clear as day.

As more advanced spacecraft have explored Mars, our understanding of the polar caps has improved, yet new data continue to reveal complex and sometimes baffling processes. We now know that underneath the seasonal CO2 caps are massive layered deposits of water ice (enough water to flood the planet 10 meters deep), which are probably vestiges of a warmer, wetter Mars. Only the south pole harbors a permanent CO2 deposit, and until recently it was thought to be a minuscule mass in comparison with the seasonal deposits. However, data from the SHARAD radar system on the Mars Reconnaissance Orbiter (MRO) revealed in 2011 that this dry ice deposit extends up to a kilometer deep, and may be enough to double the present Martian atmosphere.
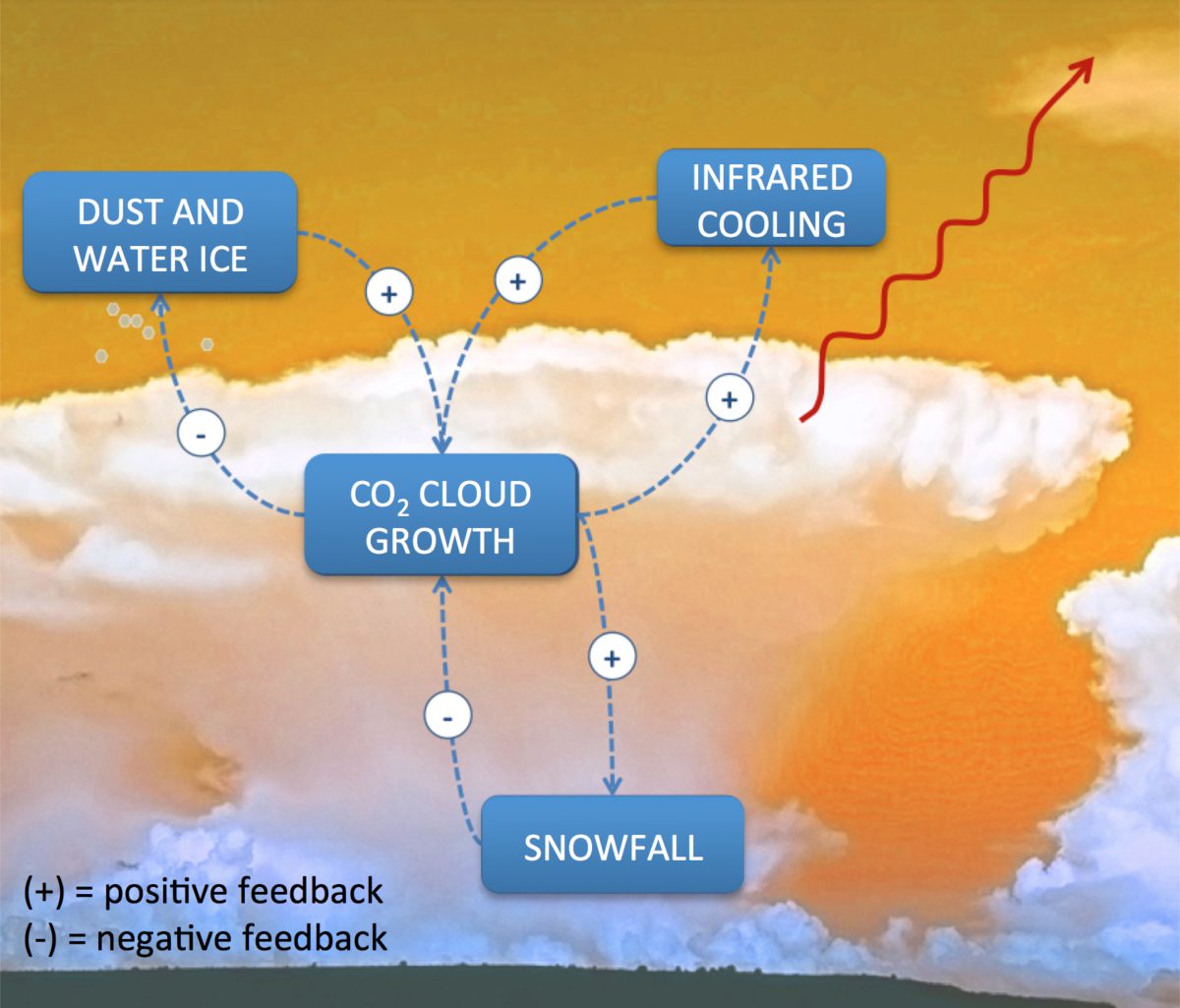
How did this massive CO2 deposit get there? Does its presence imply the atmosphere of Mars is collapsing over multi-year time scales? To the contrary, some researchers think the CO2 deposit is unstable, and may be returning to the atmosphere. They cite evidence from high resolution images taken several Mars years apart, which show the growth of "Swiss cheese" pits in the CO2 ice. Yet other observations and models suggest the cap is in a state of tenuous, but stable equilibrium. A key to settling this debate lies in the balance of energy sources and sinks in the polar regions. Chief among these are solar and infrared radiation, which compete over the energy stored as latent heat in solid CO2. In the polar winter, as on Earth, the sun is below the horizon throughout the day. Under these conditions, the infrared emission to space has little competition, and carbon dioxide condenses out of the atmosphere at a rapid rate. While the basics of this condensation process are understood, the darkness of polar night has veiled important details. Where and when does CO2 condensation occur? How rapidly is the atmosphere depleted? Does it snow?
As a resident polar scientist on the Mars Climate Sounder (MCS) science team, I led a series of two studies to investigate the possibility of CO2 snowfall and its consequences for the Martian climate. We focused on the south pole, where viewing conditions are more favorable, and the perennial CO2 cap is located. MCS has the advantage of being able to "see in the dark" with its infrared detectors, and also has its own actuators to allow scanning between the surface and atmosphere. In the first study, published in 2012 in the Journal of Geophysical Research, we used MCS infrared measurements to show that CO2 snow clouds were in fact prevalent in regions of active polar cap growth, as you can see in the image below. More than that, these clouds and snow particles were blanketing the surface by blocking infrared radiation, which strongly affects the polar energy balance. For this reason, CO2 snow clouds are important drivers of the Martian climate.

In the second study, just out in the journal Icarus, we looked at the CO2 clouds in more detail in order to estimate the rate of snowfall. Theoretical models had predicted a range from near zero, up to about 40% of the seasonal caps might be made of snow, rather than frost. We used two independent methods to test the theory. First, by retrieving vertical profiles of temperature, pressure, and CO2 ice particle concentration from the MCS data, we used models to calculate the energy budget at every level in the atmosphere and on the surface. Balancing the rate of energy loss with that released by the latent heat of CO2 deposition gives a range of 3–20% of precipitation occurring as snowfall. The second approach involved the gravitational settling of snow particles. By looking at differences in the infrared brightness of the cloud in MCS's different wavelength channels we were able to constrain the particle sizes (10 – 100 μm), which can then be fed into a model for their rate of descent through the atmosphere. This approach gave a snowfall contribution of 7–15%. With good agreement between the two approaches, we can be confident in our results. These new observations help us understand the process and the role of CO2 snowfall in forming the seasonal ice caps of Mars, and will help Mars climate scientists improve their models.
When conveying this research to the public, I often get a couple of interesting questions: "What does a Martian snowflake look like?" and "How is the skiing?" From crystallography theory, solid carbon dioxide grains are generally expected to take on a cubic form. Only a couple of experiments have been done to simulate the formation of Martian CO2 snowflakes in the laboratory, but in both cases, the researchers found the crystals took on a "cuboctahedral" shape – cubes with the corners cut off to make eight sides. These particles are much smaller (about 100x smaller) than terrestrial water snow, and lack their intricate symmetry and platy structure.

Skiing or snowboarding on Mars could be attempted on either the CO2 part of the ice cap, or the water ice part. Evidence suggests the water ice part of the cap is very ancient, and is therefore probably very dense; ice skating would be a better fit. On the fresh seasonal CO2 cap, with up to 20% made of fluffy snow, skiing might be possible. At about 1/3 the surface gravity of Earth, it would take longer to achieve a given speed on a slope, although air resistance would be negligible. "Getting air" would be much easier in the reduced gravity, so I would recommend installing a half-pipe for some insane aerials. A significant unknown is how skis or boards would perform on dry ice. Unlike on terrestrial snow, which is slippery because it typically has a thin melt layer surrounding the grains, carbon dioxide is truly "dry ice." When heated (by friction, for instance), it will sublimate straight to the gas phase. If the friction beneath a ski is too great, this could generate a layer of air, causing a total loss of control. Finally, because the CO2 snowfall occurs almost exclusively in the polar night, you had better bring your night-vision goggles.
Let’s Go Beyond The Horizon
Every success in space exploration is the result of the community of space enthusiasts, like you, who believe it is important. You can help usher in the next great era of space exploration with your gift today.
Donate Today

 Explore Worlds
Explore Worlds Find Life
Find Life Defend Earth
Defend Earth

